Lunai Bioworks Secures First Licensing LOI Following Landmark Breakthrough Showing Complete Tumor Regression in Humanized Cancer Models
Following peer-reviewed publication and independent expert review of findings, the LOI advances Lunai's tumor-regressing immune-cell platform toward scalable therapies for aggressive cancers
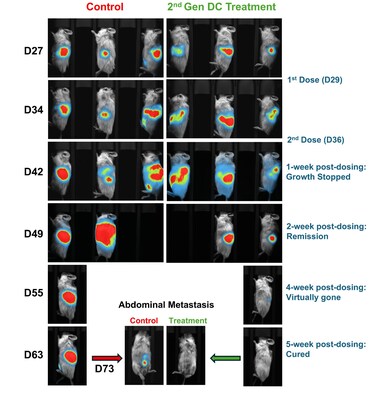
SACRAMENTO, Calif., Nov. 25, 2025 /PRNewswire/ -- Lunai Bioworks (NASDAQ:LNAI), an AI-powered drug discovery and biodefense company, today announced it has secured its first Letter of Intent (LOI) to license its next-generation immune cell therapy, which achieved complete regression of both primary and metastatic pancreatic tumors with no recurrence in humanized preclinical models.
This milestone follows Lunai's recent peer-reviewed publication in Vaccines, a successful pre-IND meeting with the U.S. Food and Drug Administration (FDA), and growing third-party recognition from the biotechnology and scientific communities. Lunai has proposed a Phase I clinical trial evaluating its Dendritic Cell Combination Therapy (DCCT) across several high-need solid tumors, including pancreatic cancer, which currently has a five-year survival rate of just 13 percent.
"We are seeing accelerating validation from both researchers and industry partners," said David Weinstein, CEO of Lunai Bioworks. "Independent expert analysis confirms the strength of our data, while early licensing activity reflects growing confidence in this platform's potential to unlock scalable, off-the-shelf treatments capable of reaching the patients who need them most."
In a widely circulated post on LinkedIn, Benjamin McLeod, Founder of Convey Bio and Co-Host of Bio2Bedside, highlighted the study as a potential breakthrough in cancer immunotherapy.
In humanized mouse models of pancreatic cancer—one of the most lethal and treatment-resistant tumors—Lunai's DCCT achieved complete regression of both primary and metastatic lesions with no recurrence. These results demonstrate potent, multi-pathway immune activation.
Additionally, the late Dr. Anahid Jewett, Professor at UCLA and a leading authority in tumor immunology commented: "In our view, these results approach what could be called the 'holy grail' of cancer research. We observed an 80–90 percent reduction in tumor size and volume across two independent studies, with most of the remaining tissue consisting of immune cells rather than cancer cells."
Lunai is also advancing additional studies and expanding clinical reach for its DCCT platform through collaborations with leading investigators, including Dr. Steven Dubinett (UCLA) for non-small cell lung cancer and Dr. Xiaolin Zi (UC Irvine) for prostate cancer.
"Lunai's dendritic cell approach has the potential to overcome longstanding barriers in solid tumor treatment," said Dr. Dubinett, Dean of the David Geffen School of Medicine at UCLA.
Lunai's DCCT introduces a first-in-class, allogeneic immunotherapy designed to scale:
- Lunai's DCCT leverages the natural antigen-presenting power of dendritic cells while eliminating the cost, time, and variability associated with patient-specific manufacturing.
- The DCCT is manufactured from healthy donor cells and stored ready-to-use. This off-the-shelf model reduces manufacturing timelines from weeks to days, lowering the overall treatment cost.
- In humanized mouse models of pancreatic cancer, one of the most lethal and treatment-resistant tumors, DCCT achieved complete regression of both primary and metastatic lesions with no recurrence.
Lunai Bioworks is preparing for formal licensing negotiations and pre-IND activities in early 2026, advancing toward clinical development of its dendritic cell therapy platform.
About Lunai Bioworks
Lunai Bioworks Inc. is an AI-powered drug discovery and biodefense company pioneering safe and responsible generative biology. With proprietary neurotoxicity datasets, advanced machine learning, and a focus on dual-use risk management, Lunai is redefining how artificial intelligence can accelerate therapeutic innovation while safeguarding society from emerging threats. For more, visit https://lunaibioworks.com.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/lunai-bioworks-secures-first-licensing-loi-following-landmark-breakthrough-showing-complete-tumor-regression-in-humanized-cancer-models-302624947.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/lunai-bioworks-secures-first-licensing-loi-following-landmark-breakthrough-showing-complete-tumor-regression-in-humanized-cancer-models-302624947.html
SOURCE Lunai Bioworks Inc.